Transgenic Animals
A transgenic animal is one that carries a foreign gene that has been deliberately inserted into its genome. The foreign gene is constructed using recombinant DNA methodology. In addition to a structural gene, the DNA usually includes other sequences to enable it
- to be incorporated into the DNA of the host and
- to be expressed correctly by the cells of the host
Transgenic sheep and goats have been produced that express foreign proteins in, for example, their milk. These animals should prove a valuable source of proteins for human therapy.
Transgenic mice have provided the tools for exploring many biological questions.
An example:
Normal mice cannot be infected with polio virus. They lack the cell-surface molecule that, in humans, serves as the receptor for the virus. So normal mice cannot serve as an inexpensive, easily-manipulated model for studying the disease.
However, transgenic mice expressing the human gene for the polio virus receptor
- can be infected by polio virus and even
- develop paralysis and other pathological changes characteristic of the disease in humans.
Two methods of producing transgenic mice are widely used:
- transforming embryonic stem cells (ES cells) growing in tissue culture with the desired DNA.
- injecting the desired gene into the pronucleus of a fertilized mouse egg
The Embryonic Stem Cell Method (Method "1")
Embryonic stem cells (ES cells) are harvested from the inner cell mass (ICM) of mouse blastocysts. They can be grown in culture and retain their full potential to produce all the cells of the mature animal, including its gametes.
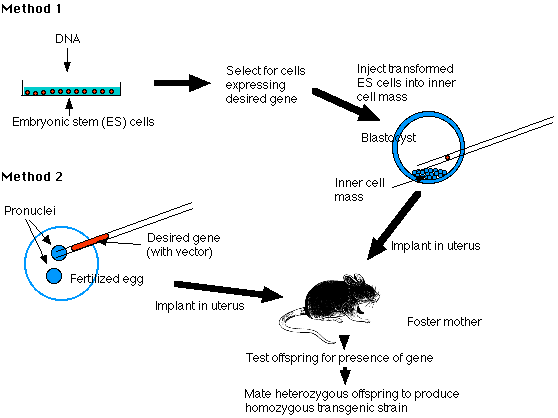
1. Make your DNA
Using recombinant DNA methods, build molecules of DNA containing
- the structural gene you desire (e.g., the insulin gene)
- vector DNA to enable the molecules to be inserted into host DNA molecules
- promoter and enhancer sequences to enable the gene to be expressed by host cells
2. Transform ES cells in culture
Expose the cultured cells to the DNA so that some will incorporate it.
3. Select for successfully transformed cells. [Method]
4. Inject these cells into the inner cell mass (ICM) of mouse blastocysts.
- Prepare a pseudopregnant mouse (by mating a female mouse with a vasectomized male). The stimulus of mating elicits the hormonal changes needed to make her uterus receptive.
- Transfer the embryos into her uterus.
- Hope that they implant successfully and develop into healthy pups (no more than one-third will).
6. Test her offspring
- remove a small piece of tissue from the tail and examine its DNA for the desired gene. No more than 10-20% will have it, and they will be heterozygous for the gene.
7. Establish a transgenic strain
- Mate two heterozygous mice and screen their offspring for the 1:4 that will be homozygous for the transgene.
- Mating these will found the transgenic strain.
The Pronucleus Method (Method "2")
1. Prepare your DNA as in Method 1
2. Transform fertilized eggs
- Harvest freshly fertilized eggs before the sperm head has become a pronucleus.
- Inject the male pronucleus with your DNA.
- When the pronuclei have fused to form the diploid zygote nucleus, allow the zygote to divide by mitosis to form a 2-cell embryo.
3. Implant the embryos in a pseudopregnant foster mother and proceed as in Method 1.
An Example
 This image (courtesy of R. L. Brinster and R. E. Hammer) shows a transgenic mouse (right) with a normal littermate (left). The giant mouse developed from a fertilized egg transformed with a recombinant DNA molecule containing:
The levels of growth hormone in the serum of some of the transgenic mice were several hundred times higher than in control mice.
This image (courtesy of R. L. Brinster and R. E. Hammer) shows a transgenic mouse (right) with a normal littermate (left). The giant mouse developed from a fertilized egg transformed with a recombinant DNA molecule containing:
The levels of growth hormone in the serum of some of the transgenic mice were several hundred times higher than in control mice.
Random vs. Targeted Gene Insertion
The early vectors used for gene insertion could, and did, place the gene (from one to 200 copies of it) anywhere in the genome. However, if you know some of the DNA sequence flanking a particular gene, it is possible to design vectors that replace that gene. The replacement gene can be one that
- restores function in a mutant animal or
- knocks out the function of a particular locus
In either case, targeted gene insertion requires
- the desired gene
- neor, a gene that encodes an enzyme that inactivates the antibiotic neomycin (and its relatives)
- tk, a gene that encodes thymidine kinase, an enzyme that phosphorylates the nucleoside analog gancyclovir. DNA polymerase fails to discriminate against the resulting nucleotide and inserts this nonfunctional nucleotide into freshly-replicating DNA.
 Treat culture of ES cells with preparation of vector DNA
Results:
Treat culture of ES cells with preparation of vector DNA
Results:
- most cells fail to take up the vector; these cells will be killed if exposed to neomycin
- in a few cells: the vector is inserted randomly in the genome. In random insertion, the entire vector, including the tk gene, is inserted into host DNA. These cells are resistant to neomycin but killed by gancyclovir.
- in still fewer cells: homologous recombination occurs. Stretches of DNA sequence in the vector find the homologous sequences in the host genome and the region between these homologous sequences replaces the equivalent region in the host DNA.
Step 2
Positive selection of transformed cells by culturing all cells in neomycin. The cells (the majority) that failed to take up the vector are killed.
Step 3
Negative selection of all cells (tk+) in which the vector was inserted randomly by culturing cells surviving Step 2 in gancyclovir. This step leaves a population of cells transformed by homologous recombination (enriched several thousand fold).
Step 4
Inject these into the inner cell mass of mouse blastocysts.
Knockout Mice: What do they teach us?
If the replacement gene (A* in the diagram) is nonfunctional (a "null" allele), mating of the heterozygous transgenic mice will produce a strain of "knockout mice" homozygous for the nonfunctional gene (both copies of the gene at that locus have been knocked out").
Knockout mice are valuable tools for discovering the function(s) of genes for which mutant strains were not previously available. Two generalizations have emerged from examining knockout mice:
- Knockout mice are often surprisingly unaffected by their deficiency. Many genes turn out not to be indispensable. The mouse genome appears to have sufficient redundancy to compensate for a single missing pair of alleles.
- Most genes are pleiotropic. They are expressed in different tissues in different ways and at different times in development.
24 June 1999

 This image (courtesy of R. L. Brinster and R. E. Hammer) shows a transgenic mouse (right) with a normal littermate (left). The giant mouse developed from a fertilized egg transformed with a recombinant DNA molecule containing:
This image (courtesy of R. L. Brinster and R. E. Hammer) shows a transgenic mouse (right) with a normal littermate (left). The giant mouse developed from a fertilized egg transformed with a recombinant DNA molecule containing:
